Watching social behaviour evolve
Evolution of bacterial cooperation observed in laboratory first-hand by Max Planck scientists
Many positive aspects of modern human society are the fruit of millennia of cooperative interactions between members of our species. Yet the evolutionary origins of cooperative behaviour in social animals and insects remains one of the most intriguing puzzles in biology. Cooperation is of evolutionary importance because it provides benefits to participants that are unavailable to isolated individuals. Starting with non-social ancestors of the soil bacterium Myxococcus xanthus, Gregory J. Velicer and Yuen-tsu N. Yu of the Max Planck Institute for Developmental Biology in Tübingen observed the real-time evolution of novel forms of cooperative behaviour between bacterial cells in the laboratory. As reported in a study appearing in the September 4 issue of Nature, the bacteria evolved the ability to cooperatively swarm across laboratory surfaces.
In addition to social animals and insects, many species of bacteria are now also known to engage in various cooperative behaviours such as the production of quorum sensing compounds and adhesive polymers that allow the formation of biofilms. Even more strikingly, the myxobacteria (to which M. xanthus belongs) are a group of particularly social bacteria. Large groups of these cells cooperate to swarm across solid surfaces in search of prey organisms which they hunt and kill together like a wolf pack. Moreover, when they encounter starvation conditions, groups of up to 100,000 individuals M. xanthus cells aggregate together to construct three-dimensional fruiting structures in which stress-resistant spores are formed.
Wild-type M. xanthus normally moves cooperatively by a mechanism known as S-motility that involves contact between cells mediated by filamentous appendages on the cell surface known as pili. Although cells can also move individually by means of an alternative motility mechanism known as A-motility, they can be forced to move socially (using S-motility) by culturing them on a soft agar surface where only S-motility is able to drive effective movement.
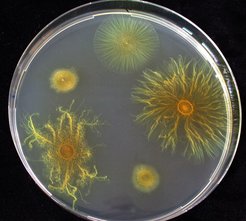
Velicer and Yu posed the question whether M. xanthus might be able to somehow re-evolve the ability to swarm socially on soft agar even if it lacked the pili that are absolutely necessary for the normal social swarming by S-motility. To examine this question, a gene encoding the primary structural component of pili was deleted from the M. xanthus genome. The resulting non-social mutant was unable to swarm effectively on soft agar and was used to initiate multiple replicate lineages that were allowed to grow and evolve on soft agar over a period of 64 weeks. Cells were inoculated onto the center of nutrient soft agar plates and every two weeks a small section from the outside edge of the growing colony was transferred to a fresh plate. After many weeks of culturing, two of the evolving populations had begun to swarm outward at a rate almost as fast as normal wild-type colonies (that have functional S-motility).
The swarming patterns of the two evolved populations, however, were very different than those of normal S-motility swarms, suggesting that they might be migrating by a novel mechanism. Velicer and Yu confirmed this by showing that the evolved populations had not regained the ability to make pili (and were therefore not moving by pili-mediated S-motility). Instead, it would appear that the propulsive force for the evolved swarming is provided by A-motility (the normal motor for individual motility) but an additional social component is required as well. The descendant populations had evolved the ability to make enhanced quantities of an adhesive polymer matrix that cause their cells to literally stick together to a much greater degree than their non-social (i.e. non-piliated) ancestors. This intercellular adhesive matrix, composed of strands called fibrils (made of carbohydrates and proteins), was demonstrated to be essential for the swarming ability of the evolved strains. Both genetic and chemical inhibition of fibril-matrix construction inhibited the evolved swarming phenotypes.
Genetic mutants of the evolved strains were constructed that are unable to make fibrils and are therefore do not swarm. The swarming defects of these mutants, however, could be rescued by the mere social presence of their fibril-producing parents. When cultured alone on a plate, the fibril-deficient mutants swarm very poorly. When mixed as a minority with their adhesive, fibril-constructing parents, they swarmed much further than they did alone due to the presence of the fibril matrix generated by the evolved parental strain.
The evolved swarming ability in this study is cooperative because it depends on the contribution of fibril material by individual cells into the public commons between cells. Just as human organizations fail to function if too few people contribute their fair share, so also the evolved swarming of M. xanthus fails if too few cells in a group contribute adhesive fibrils.
This study by Velicer & Yu demonstrates that social microorganisms provide a rich opportunity to experimentally address questions about the evolution of cooperation between individuals that have challenged theoreticians for decades. For many microbes, including pathogens, social interactions with other members of the same species are important for evolutionary success. Further research on how cooperation within such miniature societies evolves could shed light on intriguing questions and be utilised in the battle against social pathogens.












