Chaperones fold Rubisco
Photosynthesis, a veritable stroke of genius on the part of nature, makes the existence of higher life forms possible. If it can be optimized, it may be able to make an even greater contribution to the resolution of future energy problems. Manajit Hayer-Hartl and Ulrich Hartl are currently working on this possibility at the Max Planck Institute of Biochemistry in Martinsried.
Text: Harald Rösch

Be honest – when you look at a rich green meadow, does a solar power plant come to mind? Of course not. However, plants do something very similar: they convert the energy from sunlight into usable energy. Through photosynthesis, they harness this energy to synthesize sugar in a process that involves numerous intermediate steps. We already tap into this energy source today, for example in the form of biofuel and in biogas plants.
So nature has been building solar power plants for millions of years. The only drawback is that these natural power stations work so inefficiently. Plants are energy wasters – at least when it comes to harnessing energy through photosynthesis. The combined area of Germany and France would not provide sufficient acreage to cover Europe’s bioethanol or biodiesel requirements in 2050. If, however, 10 percent of the energy that falls on this area in the form of sunlight could be converted into chemical energy, an area the size of the German federal state of Baden-Württemberg would probably be sufficient.
The efficiency of plant photosynthesis is only around 5 percent. In comparison, the solar power cells available today have an efficiency of around 20 percent, but they don’t produce fuels that, like bioethanol, can be easily stored and transported. However, there are organisms that achieve far greater levels of efficiency than plants. The green sulfur bacterium Chlorobaculum tepidum, for example, has extremely efficient solar power equipment for the absorption of light, and can convert 10 percent of incident light into chemical energy.
Turbo plants harness more energy
For this reason, scientists are working on developing ways to make photosynthesis more effective. Their aim is to optimize various steps in the process of the conversion of light energy into chemical energy, for example by increasing the efficiency of the photosynthesis machinery.
Plants that have been upgraded in this way would be capable of forming more biomass for the purpose of fuel production. In contrast to this, some scientists want to dispense with plants altogether and equip bacteria with an optimized photosynthesis device instead. Another possible approach will be independent of the services of any kind of organism: hydrogen, for example, could be generated in bioreactors in which photosynthesis takes place using just a few necessary proteins. Nature has, in fact, created enzymes that can split water with the help of solar energy and that could thus replace costly platinum in fuel cells. And nature also has enzymes that subsequently produce hydrogen from the water fragments.
At the Max Planck Institute of Biochemistry in Martinsried, near Munich, researchers are looking for a way to make plant photosynthesis more effective – and have already made significant progress toward achieving this objective. The scientists working with Manajit Hayer-Hartl have discovered the folding process of a key protein in photosynthesis, known as Rubisco. Armed with this knowledge, the researchers can now work on finding a way to produce Rubisco artificially and modifying it so that it works more efficiently.
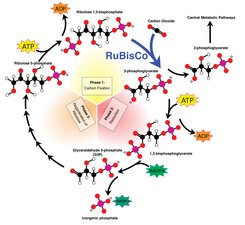
Rubisco is not only the most common protein on Earth, it is also one of the most important. Without Rubisco, life would simply not exist in its current form. It binds carbon dioxide from the atmosphere and brings about its conversion to sugar and oxygen. However, Rubisco works very slowly and ineffectively. It reacts not only with carbon dioxide, but also with oxygen: on average, it binds one oxygen molecule after three to five carbon dioxide molecules. “When Rubisco emerged around four billion years ago, this was irrelevant, as there was not yet any oxygen in the atmosphere. Today, however, the amount of oxygen in the air is around 20 percent,” says Manajit Hayer-Hartl, who is carrying out research on Rubisco with her husband Ulrich. Rubisco could, therefore, work far more effectively if it no longer reacted with oxygen.
Chaperones ensure good order and form
That is why the Martinsried-based researchers want to modify Rubisco in such a way that it can bind only carbon dioxide. To do this, they must first establish how the protein is actually formed. Rubisco is one of the largest of all proteins and consists of eight large and eight small subunits. “With so many subunits, there is a significant risk that the wrong parts of the protein will aggregate and clump together,” explains Manajit Hayer-Hartl. In order for the protein to function correctly, the amino acid chains must be correctly folded and the subunits assembled so that they form a cylinder. This complex folding process is managed by special proteins known as chaperones.

According to the researchers, three proteins are required to recreate a functional Rubisco complex: in addition to the previously identified chaperonins GroEL and GroES, a recently discovered helper protein (RbcX). RbcX ensures that two large subunits of Rubisco can assemble next to each other. Four of these dimers then form the cylinder, and four small subunits position themselves at the top and bottom areas of the cylinder. “We now understand why, for example, bacteria are not able to produce functional Rubisco. If we insert only the DNA for the protein into the bacterial genome – without the corresponding helper protein – functional Rubisco cannot be formed,” says Ulrich Hartl.
Having achieved this breakthrough, the scientists can now get to work on producing Rubisco artificially in the laboratory. To this end, they want to introduce the DNA for Rubisco, the two chaperonins and the helper protein into bacteria. The rapidly reproducing microorganisms will then produce the Rubisco protein in sufficient quantities. The researchers are seeking to find a more efficient variant of Rubisco with the help of such bacteria. “If we introduce the Rubisco DNA into a bacterial strain that can survive only with functional Rubisco, we can test all possible mutations in the Rubisco gene and immediately establish how well the individual variants work,” explains Manajit Hayer-Hartl.
Can humans succeed where nature failed?

With the help of this process, multiple mutations can also be generated and studied in different positions in the Rubisco gene. This is an important advantage, as it may not be possible to further optimize the protein by replacing a single amino acid. This would explain why nature itself did not adapt Rubisco to the increasing oxygen content in the air over the course of evolution. Some scientists believe that nature has already found the optimum structure for Rubisco, and that Rubisco cannot be improved. The scientists in Martinsried disagree. They are convinced “that the Rubisco molecule of plants is definitely not the best possible variant. Some red algae have a more efficient form. This suggests that the plant enzyme can be improved, too.”
However, finding mutations that would render Rubisco more specific to carbon dioxide is not the only challenge the scientists have to face. The new results show that nothing will work without the matching molecular chaperones. Unlike Rubisco itself, RbcX works extremely selectively and also assists in the folding of the natural plant Rubisco. For this reason, it has not been possible, for example, to transfer the red-algal Rubisco to plants – it simply does not fold correctly in this case. It is thus possible that an optimized Rubisco variant will also require its own specific assembly chaperone.
More energy with less water
Despite all the difficulties it presents, the objective behind this research is worthwhile: on the one hand, algae or plants with an optimized Rubisco variant could be used as a weapon in the fight against the rising carbon dioxide concentration in the atmosphere; on the other hand, the availability of such turbo plants with a significantly higher growth rate would also be a huge advantage for agriculture. “We could benefit from a form of Rubisco that is 10 or 15 percent more efficient,” says Manajit Hayer-Hartl.
It is not just a question of accelerating growth, but of even making it possible in the first place in some locations, as more efficient conversion of carbon dioxide into sugar reduces the plant’s water consumption. As a result, in the future, agricultural activity would be possible in areas that are currently too arid for today’s crop plants – and such areas are set to expand further due to the increasing scarcity of water on Earth.
GLOSSARY
Chaperone
Proteins can function only if their amino acid chains are correctly folded. Like the chaperones of the 19th century whose job it was to shield young ladies from improper influences, special enzymes in cells ensure that proteins do not end up on the wrong path and assume the wrong form. Some chaperones take the form of a cylinder in which only a single molecule can fold. Such chaperones found in bacteria, chloroplasts and mitochondria are known as chaperonins. A lack of functional chaperones can result in the clumping of proteins and cause various diseases, such as Alzheimer’s and Huntington’s chorea.
Photosynthesis
Photosynthesis involves the production of carbohydrates from carbon dioxide and water with the help of solar energy. The process can be subdivided into two connected stages: The light reactions (photo part) make energy available for the water to be split into electrons, protons and oxygen. The energy-rich electrons and protons are used in the Calvin cycle (synthesis part) to convert carbon dioxide into sugar.



