Vaccines from a reactor
Text: Tim Schröder
The weapons against influenza include the egg – the plain old chicken egg. The egg is, after all, a biotechnology laboratory in miniature. In 1931, pathologist Ernest W. Goodpasture at Vanderbilt University in Nashville made a momentous discovery. He pricked an incubated egg with a fine needle and infected it with influenza viruses. The viruses reproduced prodigiously in the egg. When Goodpasture drew a bit of liquid out of the egg and examined it a few days later, he found that the number of viruses had skyrocketed. Goodpasture immediately realized that eggs are an ideal host for growing influenza viruses and, since vaccines require viruses, the perfect tool for producing vaccines.
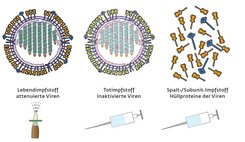
The trick is to carefully inoculate the body with viruses without making it ill. The immune system then learns to recognize the pathogen and is able to mount a defense against it. Medicine uses three basic immunization methods for this. The first is to inject a large number of killed viruses; the second is to inject smaller numbers of attenuated and therefore non-infective viruses; and the third is to inject fragments of the virus shell or individual viral proteins. In any case, all three methods require viruses.
Vaccine production needs to become more efficient
Goodpasture’s method is now 80 years old and has been continuously refined. Nevertheless, the egg remains the method of choice for manufacturing flu vaccines. Ninety-five percent of all flu vaccine doses still contain egg-grown viruses. However, this method is nearing its limits: for a single vaccine dose, one or two eggs have to be embryonated in a temperature-controlled cabinet, and many millions of eggs are needed to produce enough vaccine to supply an entire country.
In the European Union, it has so far been possible to provide a sufficient stockpile of vaccine for the next flu epidemic using this method. “But what happens if an epidemic breaks out in China or India, countries with a combined population of more than two billion? Production couldn’t keep up,” says Udo Reichl, Director of the Department of Bioprocess Engineering at the Max Planck Institute for Dynamics of Complex Technical Systems in Magdeburg.
The biologist and process engineer has been working with his team on developing alternatives to vaccine production in eggs. Like vaccine manufacturers and other research groups around the world, he is pinning his hopes on animal cells cultivated in laboratory vessels and bioreactors. However, it is expensive to replace an established method of pharmaceutical production, and the industry is very reluctant to do so. Udo Reichl is therefore seeking to make the production of vaccines in cell cultures so efficient that companies will see it as a viable alternative.
The cells that come into question for virus production were, for the most part, extracted from various animals and organs many years or even decades ago – from monkeys, hamsters and dogs, for example. Many of these cell lines are immortal, meaning that they can propagate indefinitely. There are also some new cell lines that research institutions and biotech companies have rendered genetically immortal. These, too, would be suitable for use in pharmaceutical production.
Udo Reichl’s team has identified several such cell lines that are particularly suitable for growing viruses. “It’s ironic,” says Reichl. “Other scientists are busy fighting viruses and keeping their numbers as low as possible, while we’re trying to stimulate a cell to produce as many viruses as possible. Our work isn’t anti-, but pro-viral.”
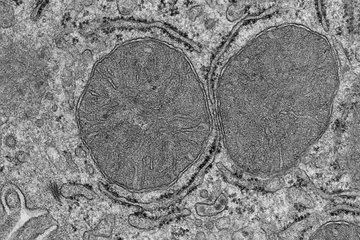
It is both fascinating and frightening how a virus infects a cell and reprograms it to release thousands of copies of itself. An influenza virus resembles a spiked ball. The spikes consist of the proteins hemagglutinin and neuraminidase. At the tip of the hemagglutinin spike is a lock-like structure that enables the virus to bond to the surface of animal or human cells. The fine structure of this site determines whether the structures on the cell surface fit the viral hemagglutinin like lock and key, thus allowing the virus to enter the cell.
The fight against influenza is a race against time
If the lock on the virus surface finds a corresponding key on the cell surface, the influenza infection begins to run its fateful course. The membrane of the host cell opens up, and the virus penetrates into the cell and releases its genome inside the nucleus. The viral RNA then reprograms the cell to act as a virus factory. The cell blithely synthesizes viral components, which are then assembled into hundreds or even thousands of new viruses. The assembled viruses bud off from the cell surface, a process that requires the viral protein neuraminidase. For humans and other animals, it is disastrous when the viruses start to replicate so prolifically – that’s when influenza really takes hold. For vaccine production, however, it is ideal.
While influenza viruses are, for the most part, still being produced in eggs, other types of viruses have long been grown in cell cultures. But the aim of vaccine developers in both cases is the same: to produce large quantities of viruses in a short time so as to have sufficient vaccine on hand in the event of an epidemic or global pandemic. Unfortunately, it is pointless to stockpile some vaccines because many viruses – particularly the influenza virus – readily mutate, giving rise to new pathogens against which the available vaccine is useless.
The fight against influenza viruses is thus a race against time. Will scientists be able to identify a new virus variant and adapt the composition of vaccines before the pathogen is able to trigger a flu epidemic? More often than not, pharmaceutical manufacturers and researchers win the race. But all too often, the viruses are quicker. Things then get tricky, because the virus may spread rapidly and trigger a pandemic. In such cases, it would be good to have a fully automated breeding machine for influenza viruses that could be ramped up quickly and churn out the viruses in large quantities – a production line such as Reichl is hoping to develop.
The basic technology for growing viruses on a large scale is already available. It uses a type of cultivation vessel known as a bioreactor. However, there are obstacles to achieving optimum virus yield. The first is that the cells used for growing viruses – such as those in embryonated chicken eggs – have to reproduce rapidly. Afterwards, they have to be washed, provided with fresh nutrient medium and then inoculated with virus seed to replicate progeny viruses.

Several cell cultivation methods are currently available. For instance, there are cells that grow and replicate only if they are able to attach to surfaces – so-called adherent cells. With these cells, the maximum quantity is severely limited, because eventually the surface available for cell attachment becomes densely covered, at which point cell division stops. By contrast, some cells grow only if they are floating freely in nutrient solution, a cell suspension. Typically, much higher cell concentrations can be reached in a suspension than with adherent cells. However, cells in suspension have a tendency to clump together, which can impede the process. In addition, clarifying the cells from the medium is a more elaborate process. A third method, a middle road, has therefore been developed for bioreactors: a kind of pseudosuspension for adherent cells in which the cells are grown on microcarriers. The microcarriers, in turn, float freely in the liquid medium.
“On average, concentrations of around two to three million cells per milliliter of medium can be achieved with this method. With the type of cultivation strategy we envision, we hope to reach concentrations that are ten to a hundred times greater. We call the method high-cell-density cultivation,” says Yvonne Genzel, team leader in Udo Reichl’s department. That is an ambitious goal, but the researchers are able to tweak the process in many ways. Being highly sensitive, cells require the right medium with the ideal composition of nutrients, including vitamins, minerals and sugars. The temperature must be set exactly right, just like the pH value and the oxygen content of the medium. A stirring mechanism is required to keep the cells or microcarriers in suspension, to prevent clumping and to distribute the nutrients and oxygen evenly.
Hollow fibers expected to boost cell yield
Few research groups in the world have the ability of the Magdeburg-based biotech researchers to manipulate all the many factors involved in the cultivation of viruses and cells – from the type of the reactor to subsequent modification of a vaccine. Reichl’s team, for example, is able to study the process in all its complexity and analyze the effects of every change in detail, allowing them to control and fine-tune the process. The laboratory in Magdeburg is equipped with a variety of glass reactors in which tiny propellers spin. “These mixers must have the right shape and run at the right speed so that we don’t damage the cells,” says Yvonne Genzel. She opens the steel door of an incubation cabinet. Inside is a vibrating platform, and fastened to it are small glass flasks containing floating cells. She removes one of the flasks and holds the brownish, cloudy solution against the light. “Quite a high cell concentration,” she notes.
For high-cell-density cultivation, the nutrient medium must initially be changed every few days, and then more and more frequently to ensure that the cells are sufficiently supplied with nutrients. Only then can they divide frequently and reach high concentrations in the solution. At this point, the oxygen supply becomes the limiting factor. The more cells there are floating in the solution, the more oxygen they consume. The researchers therefore pump pure oxygen into the vessels to supply the cells. But even this approach has limitations, as the stirrer cannot be run at a sufficient speed to distribute the oxygen uniformly throughout the highly concentrated cell culture. Nevertheless, Reichl, Genzel and their colleagues have already achieved remarkable cell concentrations. With the process considered for influenza virus production, they have already attained a concentration of 50 million cells per milliliter. In the future, they hope to raise that figure to 500 million.
A very recent, promising technique may help to further boost the cell density. Yvonne Genzel reaches for a glass tube containing a thick bundle of thin white hollow fibers. The membrane of these fibers allows oxygen and nutrients to pass through, but is impermeable to viruses and cells. With the help of the hollow fiber bundle, cells can be cultivated in a continuous flow. In this process, the cavities between the fine hollow fibers are filled with cells. Nutrient medium is then pumped through the fibers. Like arteries in the body, the nutrients and oxygen diffuse through the fiber membrane to the cells. “In this way, we’re able to achieve better distribution of nutrients and improved cell growth compared with conventional bioreactors,” says Genzel. One advantage is that it isn’t necessary to separate the spent nutrient medium from the cells and replace it – a very tedious process. All that is required is to pump fresh nutrient medium through the hollow fiber unit.
However, the scientists are still a long way from perfecting the process, because the number of virus-producing cells isn’t the only consideration. It’s also important for the cells in high-density cultures to be in good physiological condition, as only then can they produce viruses in large quantities. A high virus yield depends not only on the cell count, but also on the number of viruses each cell produces. This varies between several hundred and tens of thousands of viruses per cell.
A key factor is finding the right time to infect the cells in the culture with viruses: on the one hand, the cell count should be as high as possible; on the other hand, the cells must be fit. “It takes a lot of experience to identify the right conditions,” says Genzel. If the conditions are right, the researchers inoculate the cells with seed viruses. Seed viruses must also be in good condition and highly infectious so that each one successfully initiates intracellular virus replication. After six to eight hours, the infected cells start to release the first viruses, which, in turn, infect new cells. As the cells slowly die, billions of fresh viruses accumulate in the nutrient medium. The time at which the viruses are harvested is also critical for obtaining a high virus yield. This is particularly true for vaccines containing live viruses. The longer the virus harvest is delayed, the more likely it is that inactive, non-infectious viruses will accumulate in the reactor. The key is to determine the harvesting time at which the reactor contains a maximum number of infectious viruses. The process is less time-critical for vaccines that use inactivated viruses, because the viruses don’t have to be active. Virus cultivation in Magdeburg isn’t just a matter of process engineering, though. “We also want to gain insights into the cells and try to modify them so that they produce more viruses,” says Reichl. To do this, he has established a large array of analytical tools – a high-performance liquid chromatography system, for example, that can measure the cells’ metabolic products during viral infection and production.
Conventional methods are usually set in stone
“I would have expected the cells to crank up their metabolism while they’re producing viruses,” says Reichl, “but they act just like healthy cells. Following an infection, not much changes in the cells until most of the viruses have been released and the cell dies.” Reichl can do more than measure the cells’ metabolic products. Using flow cytometry, for instance, where one cell after another zips through a tiny glass tube, he can also count and analyze the cells.
The unit can count tens of thousands of cells per second. In addition, a laser beam aimed at the cells measures, for example, which viral proteins a cell is producing at the moment and whether the proteins are located in the cytoplasm or the nucleus of the cell. It still isn’t entirely clear how Reichl and Genzel can use this knowledge to improve virus production in cells, but one thing is certain: “Few researchers are looking as closely as we are at the fundamental processes that are critical for the growth of viruses in cell cultures for vaccine production,” says Genzel. The same can be said of vaccine manufacturers. “Companies usually can’t afford basic research. Furthermore, very few companies are experimenting with the established virus cultivation processes, as it is very expensive to modify and optimize production methods.”
“Because of very tight regulations, established methods are usually set in stone. Some manufacturers are even still using conventional adherent cells – that is, cells that require a solid surface to grow on – for cultivating viruses.” However, when one considers that the development of a new vaccine production method costs several hundred million euros, it’s clear why the industry is relatively conservative in its approach.
The market for vaccines has grown enormously
Genzel and Reichl are working not only with influenza viruses, but also with a long established virus type, the modified Vaccinia Ankara Virus (MVA), which is derived from the smallpox virus. This virus was produced over a period of many years by one of the pioneers of immune research, Munich-based veterinarian Anton Mayr. This vaccination virus is very well tolerated and non-infectious, making it highly suitable for work in simple laboratories. In contrast, any researcher wanting to work with the Ebola virus, for example, would have to meet extremely high safety standards, including the use of air locks, negative pressure systems and air filtration systems.
MVA is interesting particularly because it is a suitable transporter of genes. Such transport viruses are called vectors and are used, for example, in medicine to treat diseases caused by genetic defects. For instance, some individuals have a defect in the gene containing information for the synthesis of a metabolically important protein. Using a vector, it is possible to insert the intact working gene into the body, thus providing it with the information it needs to synthesize the protein.
Moreover, vectors can also be used to immunize the body against dangerous diseases. Researchers hope that this can also be achieved for diseases such as Ebola and HIV/AIDS. For immunization purposes, MVA would be coated with the surface proteins of a dangerous virus, for example the AIDS virus. Because MVA is harmless, the safety risk for the patient is low. The immune system, however, learns in this gentle way to recognize the surface of the dangerous virus and is then able to develop immunity against it.
During the Ebola epidemic last fall, researchers published the results of an experiment in which they vaccinated macaque monkeys with an MVA vector carrying an Ebola protein. The study results caused quite a stir, as a positive test in macaques evidently means that immunity against Ebola can be achieved in apes.
Ebola is a disease that repeatedly makes headlines despite the fact that its death toll pales in significance to that of influenza, malaria and tuberculosis. “Consequently, the industry has little interest in the costly process of developing Ebola vaccines,” says Genzel. She hopes to be able to contribute to the development of inexpensive vaccine production with the help of an efficient technical process. That is certainly conceivable, because the market for vaccines has grown enormously in recent years, not least due to the frequent occurrence of bird and swine flu. In 2001, sales of vaccines totaled USD 6.9 billion. In 2009, that figure grew to USD 25.2 billion, and for 2015, it is expected to reach USD 56 to 64.2 billion.
Nevertheless, the researchers in Magdeburg still have a lot of work to do. “A virus looks simple, but it is extraordinarily adaptable and, to a certain extent, unpredictable,” says Reichl. “An influenza virus has only eight gene units that code for a dozen or so proteins. I used to think that it must be easy to understand such a virus, but that’s not the case.”
One uncertainty stems from sugar structures on the surface of hemagglutinin, which are key factors in determining the virulence of the virus. For instance, it has been known for some years that many proteins travel through the body, not in pure form, but with attached sugar groups. Experts call this phenomenon glycosylation.
For Reichl, this means: “If we want to better understand the replication of viruses and the effects of vaccines, we also have to investigate the glycosylation of their proteins.” And glycosylation means delving down to the molecular level. “Fortunately, we have the equipment and expertise at the institute to analyze the structure and composition of these sugars.”
Reichl knows that the fight against viruses will never be won. “There are too many viruses around the corner. But if we can manage to produce the right vaccine in large quantities, we will already have achieved a great deal.” As far as the influenza virus is concerned, he has another reason for exploring new technological processes and finding alternatives to the egg: If a bird flu spirals into a pandemic, things could get tricky. “If we have to slaughter the stock of poultry to stop the virus, how in the world are we supposed to obtain eggs to produce vaccines?”
To the point
Established production methods for vaccines that use embryonated chicken eggs would probably be unable to meet demand during a global flu pandemic.
Researchers from the Max Planck Institute for Complex Technical Systems are therefore looking at ways to produce large quantities of influenza vaccines, for example in animal cell cultures.
In order to increase the concentration of animal cells in a nutrient solution and obtain as many viruses for vaccine production as possible, the Magdeburg-based scientists are systematically varying all the factors involved in cell cultivation. They are also looking for the optimum time to harvest the viruses from the infected cell culture so as to ensure maximum yield.
Max Planck researchers are able to produce not only influenza viruses, but also MVA viruses, making the method interesting for vaccines against Ebola and HIV, as well.