Alternative fuel for string-shaped motors in cells
A unique two-component molecular motor uses a kind of renewable chemical energy to pull vesicles toward membrane-bound organelles
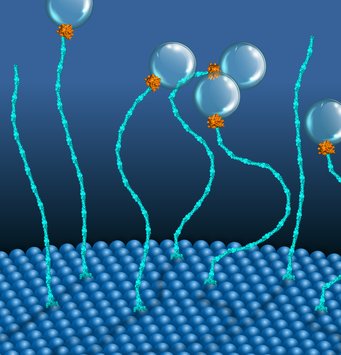
Cells have a fascinating feature of organizing their interior neatly by using tiny protein machines called molecular motors that generate directed movements. Most of these machines run on ATP, a common fuel in the form of chemical energy. Researchers from the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG), the Cluster of Excellence Physics of Life (PoL) and the Biotechnology Center (BIOTEC) of the TU Dresden in Dresden, Germany, and the National Centre for Biological Sciences (NCBS) in Bangalore, India, discovered a new molecular motor that uses an alternative chemical energy, GTP, and a novel mechanism to perform mechanical work. By repeatedly contracting and expanding, this molecular motor functions similarly to a classical Stirling engine and helps to distribute cargo to membrane-bound organelles. It is the first motor to use two components, two differently sized proteins, Rab5 and EEA1, and is driven by GTP instead of ATP.
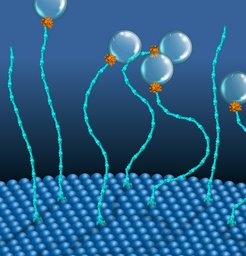
Motor proteins are remarkable molecular machines inside the cell that convert chemical energy into mechanical work. The most prominent example is myosin, which helps our muscles move. Most molecular motors use ATP as a source of energy. Now, an interdisciplinary team of cell biologists and biophysicists in the groups of MPI-CBG directors Marino Zerial and Stephan Grill and their colleagues, including PoL and BIOTEC research group leader Marcus Jahnel, has found that a small protein called Rab5 uses GTP, a different energy-rich molecule, to generate molecular forces in the cell. Additionally, it does this in cooperation with a second protein, EEA1, forming a previously unknown two-component molecular motor system.
In an earlier study, this team found that the small globular GTPase Rab5 binds to the long rod-shaped protein EEA1 and causes a contraction in EEA1. Usually, EEA1 is rather rigid, but Rab5 binding sends a message along the elongated structure of EEA1, thereby increasing its flexibility, similar to how cooking softens spaghetti. The flexibility change then produces a force that moves cargo within the cell. Now the question arose, was this a one-time reaction or could the proteins generate this movement in continuous cycles like a motor using the energy of GTP?
This is where the current research sets in, taking shape via the doctoral work of the two first authors of the study. Joan Antoni Soler from Marino Zerial's research group at MPI-CBG and Anupam Singh from the group of Shashi Thutupalli, a biophysicist at the Simons Centre for the Study of Living Machines at the NCBS in Bangalore, set out to experimentally observe this motor in action.
Modification of EEA1
With an experimental design to investigate the dynamics of the EEA1 protein in mind, Anupam Singh spent three months at the MPI-CBG. "When I met Joan, I explained to him the idea of measuring the protein dynamics of EEA1. But these experiments required difficult modifications to the protein with fluorescent probes that allowed flexibility measurement based on the protein’s structural changes,” says Anupam. Joan Antoni Soler's expertise in protein biochemistry was a perfect fit to tackle this problem. "I was delighted to learn that the approach to characterizing the EEA1 protein could answer whether EEA1 and Rab5 form a two-component motor, as previously suspected. I realized that the difficulties in obtaining the correct molecules could be solved by modifying the EEA1 protein to allow fluorophores to attach to specific protein regions. This modification would make it easier to characterize the protein structure and the changes that can occur when it interacts with Rab5," explains Joan Antoni.
Armed with the suitable fluorescent protein molecules and the valuable support of co-author Janelle Lauer, a senior postdoctoral researcher in Marino Zerial's research group, Joan and Anupam were able to characterize the dynamics of EEA1 thoroughly using the advanced laser scanning microscopes provided by the light microscopy facilities at the MPI-CBG and the NCBS. Strikingly, they discovered that the EEA1 protein could undergo multiple flexibility transition cycles, from rigid to flexible and back again, driven solely by the chemical energy released by its interaction with the GTPase Rab5. These experiments showed that EEA1 and Rab5 form a GTP-fueled two-component motor. To interpret the results, Marcus Jahnel, one of the corresponding authors and research group leader at PoL and BIOTEC, developed a new physical model to describe the coupling between chemical and mechanical steps in the motor cycle. Together with Stephan Grill and Shashi Thutupalli, the biophysicists were also able to calculate the thermodynamic efficiency of the new motor system, which is comparable to that of conventional ATP-driven motor proteins.
New class of molecular motors
"Our results show that the proteins EEA1 and Rab5 work together as a two-component molecular motor system that can transfer chemical energy into mechanical work. As a result, they can play active mechanical roles in membrane trafficking. It is possible that the force-generating molecular motor mechanism may be conserved across other molecules and used by several other cellular compartments," Marino Zerial summarizes the study. Marcus Jahnel adds: "I am delighted that we could finally test the idea of an EEA1-Rab5 motor. It's great to see it confirmed by these new experiments. Most molecular motors use a common type of cellular fuel called ATP. Small GTPases consume another type of fuel, GTP, and have been thought of mainly as signaling molecules. That they can also drive a molecular system to generate forces and move things around puts these abundant molecules in an interesting new light." Stephan Grill is equally excited: "It's a new class of molecular motors! This one doesn't move around like the kinesin motor that transports cargo along microtubules but performs work while staying in place. It's a bit like the tentacles of an octopus."
"The model we used is inspired by that of the classical Stirling engine cycle. While the traditional Stirling engine generates mechanical work by expanding and compressing gas, the two-component motor described uses proteins as the working substrate, with protein flexibility changes resulting in force generation. As a result, this type of mechanism opens up new possibilities for the development of synthetic protein engines," adds Shashi Thutupalli. Overall, the authors hope that this new interdisciplinary study could open new research avenues in both molecular cell biology and biophysics.