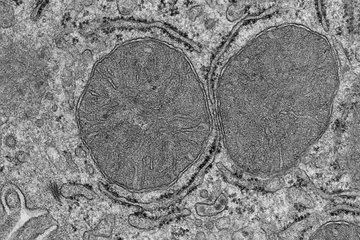
Thicker nerve fibres enable faster reactions in mice
Researchers increase the speed of signal transmission along nerve fibres in mice by switching off a protein
In order for organisms to react swiftly to stimuli in their environment, they need rapid and precise transmission of nerve impulses along neural extensions known as axons. Whereas some invertebrates have developed very thick axons to increase the neural transmission speed, in vertebrate nervous systems a myelin sheath has formed around the axon. This insulating layer enables rapid stimulus conduction with a small axon diameter. Until now, however, no-one has identified a molecule that limits axon thickness. Researchers at the Max Planck Institute for Experimental Medicine in Göttingen have succeeded in genetically switching off the CMTM6 protein in myelin-producing cells in mice. They subsequently observed that the mice not only developed thicker axons, but also reacted significantly more quickly to sensory stimuli compared to their peers. Since neurological diseases such as Charcot-Marie-Tooth disease exhibit reduced axon diameter, CMTM6 could present a possible target for therapies.

In order to flee from a predator or to hunt successfully, animals have to be able to react swiftly to external stimuli. The nerve cells conduct electrical impulses along their extensions – the axons – culminating in a contraction of the muscles. One of the factors influencing the speed with which this signal transmission occurs is the diameter of the axons. Thicker axons offer less resistance to the electrical signal, enabling swifter transmission. Invertebrates such as squid have some very large axons which achieve high transmissions speeds.
By contrast, the speed of signal transmission in the vertebrate nervous system has increased by a different evolutionary process. Myelin formed, surrounding and electrically insulating the nerve fibres with a multi-layered, fat-rich sheath. Myelin enables a fast stimulus conduction even in axons with a small diameter.
There are many reasons why this has an advantage for vertebrates: smaller axons not only use less energy, they also need less space. More axons can fit into one nerve path, enabling a greater precision in signal conduction and the development of complex cognitive functions. Insulating the axons also enables rapid signal transmission over long ranges, such as in larger animals like the giraffe. The myelin-forming cells in the peripheral nervous system, known as Schwann cells, are wound tightly around the axons. Whether this interaction limits the diameter of the axons, and to what extent, has been unclear until now.
Unknown protein in Schwann cells
Hauke Werner, along with a working group from the Max Planck Institute for Experimental Medicine in Göttingen are researching the link between myelin and axon function. Together with researchers at the University of Göttingen, the University of Leipzig, the University of Mainz and the Max Planck Institute for Medical Research, they have been researching a previously unknown protein in the Schwann cells, which sits in direct contact with the axons.
In their study, the researchers genetically switched off protein CMTM6 (chemokine-like factor-like MARVEL-transmembrane domain-containing family member-6) in the Schwann cells of mice. As a result, the mice developed significantly thicker axons than their peers. Electrophysiological investigations of the nerve cells showed an increased neural transmission speed in the mice. Other components of the nervous system were unaffected, leading the researchers to conclude that the increase in signal transmission speed could be attributed to the thicker axons.
Faster reactions
The researchers also observed faster reactions in the behaviour of the mice. “When we put the mice on a heated tile, the mice with thicker axons reacted significantly faster to the heat stimulus,” explains Maria Eichel, a colleague of Hauke Werner and first author of the study. However, the swifter signal transmission also caused problems for the mice. When the researchers set the mice to run over a grating, the genetically modified mice slipped on the bars more often than their peers. “The animals were probably experiencing problems correctly coordinating the rapidly-transmitted stimuli,” explains Eichel.
While switching off the CMTM6 protein did not create super-mice, the experiments with these genetically modified animals do however deliver a possible foundation for therapies for neuronal diseases. Many of these diseases are characterised by impaired signal transmission. In patients with Charcot-Marie-Tooth disease, reduced axon diameter is one cause. “The CMTM6 protein could be a starting point for therapies for diseases such as this,” says Eichel. The researchers’ next step will be to take mice which develop particular neurological diseases as a result of genetic modification, and also switch off the CMTM6 protein, to investigate whether their condition is improved.