A new molecular switch for cell division
The nucleic acid building block CTP regulates bacterial segregation
Before cells divide, they double their genetic material and the two copies are distributed to the daughter cells. This process sounds simple but it actually requires a smoothly functioning machinery consisting of numerous proteins.
According to scientists from the Philipps University of Marburg, the "LOEWE" Centre for Synthetic Microbiology (SYNMIKRO) and the Max Planck Institute for Terrestrial Microbiology in Marburg a newly discovered molecular mechanism prepares the cell division of bacteria by regulating the distribution of the genetic material to the daughter cells. "Our results add a new dimension to the commonly known picture," says the Marburg microbiologist Martin Thanbichler, lead author of the new study. "Apparently, the mechanisms of DNA distribution in bacteria are more complex than previously assumed.”
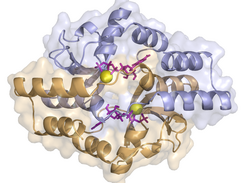
Many proteins act as switches that control important life processes in cells, including the distribution of genetic material. In order to have both DNA copies reliably transported to their destinations being located in opposite directions, various proteins bind to the genome. Researchers led by Martin Thanbichler and the Marburg biochemist Gert Bange have now investigated a central component of this machinery in more detail: they elucidated the structure of the protein ParB, analysed the behaviour of its segments and examined its mode of action within the living cell.
The scientists were surprised when it became apparent that ParB and its relatives interact with the nucleic acid building block CTP - and not with ATP or GTP, as is usually the case. "If CTP binds to a specific protein segment, two ParB molecules form a ring structure surrounding the bacterial DNA, enabling its distribution," explained Thanbichler. "The subsequent cleavage of CTP causes ParB to turn into an open form that later on can leave the DNA again.”
"ParB and its relatives can be found in almost unchanged form in many bacteria," explains Thanbichler's colleague Manuel Osorio-Valeriano, first author of the scientific paper. "Our data show that they represent a completely new class of proteins." As Thanbichler adds, " this discovery opens up completely new perspectives for research into the mechanisms underlying the vital process of DNA distribution during cell division. It will also be interesting to see whether similar CTP-dependent molecular switches play a role in other biological processes."











