Emmanuelle Charpentier: An artist in gene editing
For Emmanuelle Charpentier, deciphering the functioning of an enzyme previously known only to experts was a life-changing moment
Sometimes a single discovery can change a whole life. For Emmanuelle Charpentier, deciphering the functioning of an enzyme previously known only to experts was such a moment. The trio comprised of one enzyme and two RNA molecules and known as CRISPR-Cas made headlines far beyond the world of science. Since then, a lot of things have changed in the French woman’s life. She became a Director at the Berlin-based Max Planck Institute for Infection Biology in early October 2015.

It‘s one of the last hot days of summer 2015. Emmanuelle Charpentier sits in her office in Braunschweig, deep in concentration. She taps her foot. The dainty French woman is on the move again: after two years at the Helmholtz Centre for Infection Research, she is now moving to the German capital. In Berlin, she will be appointed Director at the Max Planck Institute for Infection Biology.
Her new institute stands on historic ground in the middle of the city: on the grounds of Charité, Berlin’s renowned teaching hospital. This is where Robert Koch, at the Prussian Institute for Infectious Diseases, began his battle against the tuberculosis bacterium at the end of the 19th century – an appropriate location for a microbiologist and one of the most important scientists of our time. For Charpentier, it wasn’t just the temperatures that made it a hot summer. She seems a little stressed from all the fuss that has surrounded her and her discovery over the last two years. The list of prizes she was awarded in 2015 alone includes several prestigious science awards, among them the Princess of Asturias Award for Scientific and Technical Research, the Louis Jeantet Prize for Medicine, the Ernst Jung Prize for Medicine, the 2015 Breakthrough Prize in Life Sciences and the 2016 Leibniz Prize.
A scalpel for researchers
As the many awards and prizes show, Charpentier’s discovery is one of science’s most remarkable success stories. The main protagonist in the story is part of the immune system of bacteria and goes by the unpronounceable name of CRISPR-Cas9. When viruses attack a bacterium, enzymes cut out a piece of the viral DNA and integrate it at a very specific location in the bacterial genome: the CRISPR sequence. The cell then transcribes this sequence into an RNA molecule, the CRISPR-RNA. The protein Cas9 then cuts the genome of invading viruses, thus incapacitating the pathogens.
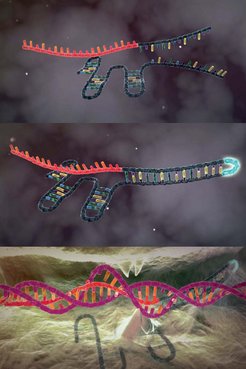
In 2011, Charpentier described the new mechanism in nature, showing that three components are involved in the process: two RNA molecules – known as CRISPR-RNA and tracrRNA – and an enzyme initially named Csn1 and now known as Cas9. In 2012, Charpentier and her colleagues published a paper in Science outlining their discovery that tracrRNA is also involved in the DNA cleavage process. Cas9 is guided by the two RNA molecules to cut the viral DNA at the location defined by the CRISPR-RNA. Emmanuelle Charpentier had already foreseen that her discovery could open up new ways of specifically targeting genes and treating human genetic disorders.
Charpentier’s group in Umeå, Sweden worked with Jennifer Doudna’s group in Berkeley, California to combine the two RNA molecules into one molecule. This step simplified the application of the CRISPR-Cas9 tool, since it meant that researchers only needed to program one RNA molecule in the gene sequence that Cas9 was to cleave.
In order for the DNA to be cut at a desired location, only the relevant gene sequence needs to be known and a matching RNA molecule manufactured. Researchers can then insert a new DNA snippet at the cut and thus switch genes on or off.
Since the discovery of the CRISPR-Cas9 mechanism, the molecular scissors have taken laboratories by storm: in 2012, 127 publications covered the subject of CRISPR-Cas9; by 2013 the figure had risen to 277. In 2015, the number skyrocketed to almost 500, and could rise to more than 1,100 studies this year. Scientists from all over the world are now taking a closer look at CRISPR-Cas9 and want to develop it further. But they are using it mainly as a tool to study genes. As it is much more precise, efficient and cost effective than previous methods, it is already a vital addition to many labs.
To date, all experiments involving plant, animal and human cells have been successful. CRISPR-Cas9 therefore has the potential for an extremely diverse range of applications: from plant breeding and the breeding of transgenic laboratory mice to treatments for a multitude of diseases. Doctors could use it to correct mutations and cure genetic diseases. It is already being used in HIV and malaria research.
An immune mechanism in bacteria, until recently known only to hardcore microbiologists, could soon become a medical treatment for millions of patients. CRISPR-Cas9 is therefore a prime example of one of Charpentier’s core beliefs: “Basic research is essential for progress,” she stresses at every opportunity – a view that resonates perfectly with the Max Planck Society, of course.
The driving force behind Charpentier’s research is, above all, an unquenchable curiosity about the world and its complex relationships, a characteristic that she displayed even as a child. Charpentier grew up south of Paris, where she was exposed to a multifaceted range of interests. Her mother worked in psychiatry; her father was responsible for planning green spaces in the city. One of her sisters began her studies at university just as Emmanuelle started school, and from then on, one thing was clear to her: she wanted that, too. Even more so than school, university seemed to her to be the place of learning and intellectual debate.
From music to biology
That she would one day be a biologist, however, wasn’t determined until later. “According to my mother, when I was 11 or 12 years old, I told her that I would work at the Pasteur Institute – and that is where I actually did my doctorate,” she recalls with a smile. “But I actually enjoyed all my subjects at school. So I could have also followed a very different path.”

In her spare time, Charpentier enjoys art, music and dance. She played the piano and practiced ballet and modern dance for several years – a good exercise, not least for discipline and creative power. As she herself says: “Art has had a significant influence on my scientific career: you need to be rigorous, but you also need to be able to let yourself go.”
But she didn’t want to make a career out of this talent: not only because it is difficult to make a living from it, but, for dancers in particular, their active years are soon over. Scientists, on the other hand, can always keep making progress, even as they get older.
Biology, with its complexity and direct relationship with people, was the area that fascinated her most. Biochemistry was the area she initially focused on, but bacteria soon took over the main role in her life as a researcher. In her doctoral thesis, she examined mechanisms that lead bacteria to develop resistance to antibiotics. It was already apparent back then that the number of multi-resistant pathogens would rapidly increase.
At the same time, there was a growing awareness that we could also learn a lot from them. Thus, the research that was conducted on bacteria in the 1970s resulted in important new laboratory techniques, such as the gene cloning.
It is surprises like these that she finds so exciting in her discipline. This is also what motivated her at an early stage, following her studies and her doctoral work, to continuously embark on new adventures – both geographically and in terms of subject matter. She began her scientific pilgrimage with a postdoctoral position in New York. On the day she arrived, she found out that her working group would be moving to Memphis, Tennessee. “At that moment, I remembered something that my aunt, a missionary, had once predicted for me as a young child: that I would have an adventurous life with constant change. Up to then, it didn’t mean very much to me. But since my arrival in New York, I have actually been constantly on the move.”
This is also a source of inspiration for Charpentier. After all, leaving different places also means constantly leaving her comfort zone, scrutinizing and tweaking her own work. “This is probably exactly what I need for my work,” she says, also recommending it to her students. Similarly, she didn’t want to restrict herself in terms of subject matter, even if that was sometimes viewed critically by scientific colleagues. After all, in the scientific world, it’s important to find one’s niche.
Leaving home initially also meant leaving her bacteria. Charpentier turned instead to their hosts, and carried out research on mice, focusing on skin development and how microorganisms trigger skin infections. In doing so, she learned how arduous it was to modify the mouse genome in such a way as to make the animals suitable as model systems for diseases. This experience ultimately brought the researcher back to bacteria, as they were frequently a starting point for new gene manipulation tools.
Back in Europe
After six years of postdoctoral work in the US, she was offered the opportunity to establish her own research group in the Biocenter at the University of Vienna. This was a real incentive to return to Europe. “Vienna offered strong basic research and outstanding colleagues, and I could choose my own subjects and work completely independently. I learned to think on a larger scale, to apply for research funding, as well as to manage with scant resources.”
In 2009, Charpentier moved from Vienna to Umeå University in Sweden. More than a few people thought she was a bit crazy at the time. “The move to Umeå was certainly risky,” she admits. “But ultimately it was exactly the right decision.” Not least because she came up with the radical idea of bringing together CRISPR and RNA while on the plane during her initial commutes between the two locations.
It then took her almost a year to find a student who also wanted to implement her idea in the lab. Her master’s student Elitza Deltcheva played an important role in encouraging other colleagues in the group to take an interest in CRISPR-Cas9.
Charpentier has now been researching in Germany since 2013. The country has always been a presence in her life. Her parents experienced the Second World War, she herself learned German at school, and as part of a student exchange, she got a taste of life in the country by living with a German family in a small town in the Rhine region for two weeks. Many of her friends and colleagues in the US, Austria and Sweden were also from Germany.
The Helmholtz Centre in Braunschweig and the Hannover Medical School enticed her with, among other things, an Alexander von Humboldt professorship. The connection with the Hannover Medical School gave Charpentier access to clinical practice – a connection that she would now like to build on in a similar way at Charité. But she still stays in touch with her colleagues in Hanover.
Legal framework absent in many countries
In addition, two of the companies that she co-founded are driving the development of CRISPR-Cas9: CRISPR Therapeutics was set up to make the technology commercially viable as a method of treatment for genetic diseases. The company wants to remove cells from patients, treat them with CRISPR-Cas9, and then return them to the body. Alternatively, CRISPR-Cas9 could also be inserted into the body via specific transport containers, such as fat bubbles or nanoparticles. Charpentier also helped co-found ERS Genomics, and has transferred the licenses for use on other organisms to the company.

However, it will still be a few years before humans can be treated with CRISPR-Cas9. Before that, more clarity about potential risks is needed. Although CRISPR-Cas9 is considerably more precise than other techniques, it still makes mistakes and occasionally cuts the genome at the wrong place.
Added to this are ethical issues that the use of CRISPR-Cas9 could raise. Chinese researchers recently modified the genes in human embryonic stem cells. While the embryos weren’t viable, the experiments nevertheless show that society and policy makers urgently need to dictate what is and isn’t allowed. “CRISPR-Cas9 can deliver huge benefits to humanity, but of course we need to handle it responsibly. Interventions into the human germline, for instance, which would influence the genome of future generations, are something that I and most of my colleagues refuse to do,” clarifies Charpentier. Ultimately, it is the responsibility of policy makers to ensure that the enormous potential of the DNA scissors isn’t abused to create designer babies.
Charpentier is looking forward to the new challenge at the Max Planck Institute in Berlin. The location offers the ideal conditions for her research: “I have always looked for a place where I can freely develop my ideas and practice science under excellent conditions. And I have always dreamed of working in a major city like Paris, London or Berlin.” In particular, the unconventional spirit of the city on the Spree River resembles her own, and could inspire her research.
For the time being, however, the daily commute to the institute may well have to suffice, as she will have very little time to enjoy the cultural life of Berlin in the coming months. After all, it’s full steam ahead for the establishment of her new “Regulation in Infection Biology” department, and it’s something that will require a huge amount of energy: labs will be modified, and some of her colleagues who are coming with her from the Helmholtz Centre will be busy setting up the labs so that the research can continue as seamlessly as possible. At the same time, she also wants to cultivate contacts with colleagues from the neigh- boring institutes of the two universities in Berlin and Charité.
In terms of subject matter, understanding regulation mechanisms in pathogens will continue to be the focus of her attention. First and foremost, she wants to finish what she started with CRISPR-Cas9. Her main objective is to gain an even better understanding of the biochemistry, specificity and efficiency of the system: “We can see that it works and we understand the fundamentals, but we’re still studying the details of how the DNA sequences are identified and how the cutting works.” She also wants to investigate other CRISPR-Cas systems, as there may be other cutting tools in the realm of bacteria that are even more suitable for studying genes.
On top of all that, Charpentier wants to strengthen the field of microbiology at the institute and attract excellent junior scientists to this biological discipline. This is also a matter of urgency, as many microbiologists will be retiring in the coming years. Compounding this is the fact that microbiology isn’t exactly seen as a discipline with a great future. Charpentier is convinced this is an error of judgment – one that she hopes to correct.
Text: Uta Deffke
Glossary
CRISPR-Cas9
CRISPR stands for “Clustered Regularly Interspaced Short Palindromic Repeats” and describes a genome sequence in bacteria. Cas9 is an endonuclease – an enzyme that cuts DNA. In viral infections, the bacteria cut sequences out of the viral genome and insert them into the CRISPR sequence. The bacteria can use the resulting transcribed CRISPR-RNA and an additional RNA molecule to identify the viral genome if it attacks again. They can cut through it, incapacitating the pathogens. In this way, the CRISPR-Cas9 system provides the bacterial immune system with a kind of memory.
RNA
The DNA molecule contains the assembly instructions for all proteins in an organism. These instructions aren’t translated directly into proteins, but rather are first transcribed into individual, much shorter RNA molecules. The chemical structure of RNA, which is a single-stranded nucleic acid, is somewhat different from that of DNA. There are various types of RNA molecules: some serve as a template for the production of proteins, while others control gene activity.














