Nearly Unbreakable
Max Planck Scientists discover a novel construction principle at the nanoscale which prevents bones from breaking at excessive force
Scientists at the Max Planck Institute of Colloids and Interfaces have found a new design principle at the nanoscale which is responsible for the enormous stability and deformabilty of bone. They found that a piece of bone stretches more than the fibres and much more than the mineral it is composed of. The scientists applied a novel technique based on the use of a brilliant beam of X-ray photons at the European Synchrotron Radiation Facility in Grenoble, France. The same bone tissue may deform differently at different scales because of the shearing of a thin glue layer between the fibres that make up bone, as well as between the tiny mineral particles that lie inside a fibre. This construction allows bones to sustain large strains without breaking, despite being made of essentially rigid units at the molecular level (PNAS, November 9, 2006).
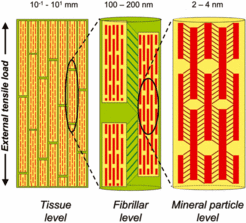
The bones in vertebrate skeletal systems need to have two main properties which at first appear to be contradictory. First, they must be rigid - a limb bone should not flop over under the weight of the bearer - and secondly, they must be tough and energy absorbing - a bone should not splinter into small fragments when dealt a blow. Usually, substances like rubber that can absorb a lot of energy by molecular conformational changes are, by the same mechanism, easy to stretch. Conversely, materials like ceramics are very rigid due to tight ionic bonds between atoms, but if the bonds break locally, runaway cracks can split the material in a brittle manner. Now in bone, half (by volume) is a stretchable fibrous protein called collagen and the other half a brittle mineral phase called apatite. By as yet incompletely understood construction strategies, such biomineralized tissues exhibit remarkably high strength and toughness necessary for their physiological functioning.
The key to understanding how this optimal construction is achieved lies in the hierarchical arrangement of organic and inorganic molecules from the nanoscale (1 level up from atomic level) up to the micron level. Using a very bright beam of photons emitted relativistically from an accelerated stream of electrons, scientists at the Max Planck Institute of Colloids and Interfaces were able to generate - in real time - diffraction image sequences of molecular and supramolecular rearrangements in bone, under applied stress. They then used these diffraction spectra as quantitative strain measures at the atomic and higher levels to build up a hierarchical picture of bone deformation.
The scientists found that the hierarchical structure of bone leads to a hierarchical deformation, as shown in Figure 1 below. Specifically, a unit of strain applied to the whole bone is absorbed by soft layers at successively lower length scales, until less than a fifth of that strain unit is actually seen by the mineral phase. Essentially, from the atomic to the micrometer level, bone consists of rigid units joined together by a soft phase, where most of the deformation occurs. These composite structures form a single rigid unit at the next level and so on, enabling the tissue to sustain large strains despite being comprised of essentially not deformable particles at the atomic level. The brittle apatite phase is thus shielded from excessive loads, and does not break.
A second unexpected finding was that the tiny mineral crystallites are nonetheless very strong, capable of carrying more than 2 - 3 times the breakage load of bulk apatite. Essentially, the small size of the particles means they cannot contain any large cracks. Since their strength is hence not "defect - limited", it can approach the values for a perfect crystal. While the existence of such a reinforcing effect is known in composite materials science, this is the first experimental evidence for this effect in biomaterials - small particles resist failure better. The natural design principles quantitatively observed here in bone - hierarchical deformation, matrix sensitivity, and nanoscale strengthening - may provide guidelines for the development of bio-inspired and new nanocomposite materials as well. Furthermore it may help bone biologists to understand how a molecular level change can cause whole bones to become more prone to fracture in diseases like osteoporosis.
