miR1 and miR133a block division of heart cells
Approach for strengthening heart regeneration discovered
The heart consists of many individual heart muscle cells that are electrically interconnected and represent a functional unit. New cells created by cell division would disrupt this interconnection and reduce the performance of the heart. Scientists from the Max Planck Institute for Heart and Lung Research in Bad Nauheim have now discovered that the two micro RNA molecules miR-1 and miR-133 in the adult heart inactivate two genes that normally force cell division. If these molecules are switched off, the heart was less damaged after a heart attack in experiments with mice. In the future, miR-1 and miR-133 could be used to specifically switch on cell division in the heart, for example to stimulate heart muscle regeneration after a heart attack.

The mammalian heart is a high-performance organ. Reliably and adapted to the respective demand, it pumps blood through the body at a relatively high pressure. It is assumed that in the course of evolution this performance capacity was bought by the fact that the organ's ability to regenerate was largely lost. The reason for this could be that the individual heart muscle cells are permanently and very closely connected to form a so-called electrical syncytium. The individual cells are interconnected via cell-cell contacts so that the electrical excitation conduction necessary for heart contraction can take place in a controlled manner. New heart muscle cells produced by cell division could disrupt this physiological process and cause the heart to become out of sync. Accordingly, in contrast to organs with a high regenerative capacity, no divisible, regenerative stem cells are found in the heart. The disadvantage: if parts of the heart muscle are damaged, as is the case with a heart attack, for example, the heart lacks the self-healing powers. Functional regeneration of the damaged heart therefore does not usually take place.
It can be assumed that the suppression of cell division in heart muscle cells is regulated in various ways. Scientists from Thomas Braun's department at the Max Planck Institute for Heart and Lung Research in Bad Nauheim have now been able to show in mice that one such mechanism involves two so-called micro-RNAs, miR-1 and miR-133a. "Micro-RNAs are short, non-coding RNA molecules that are crucially involved in regulating the activity of genes. We found that miR-1/133a suppresses two receptors on heart muscle cells. The two molecules, oncostatin M receptor and FGF receptor 1, control the division activity of heart muscle cells in the adult heart," says Thomas Böttger, one of the lead authors of the study.
To investigate the background in more detail, the Max Planck researchers used heart muscle cells in which miR-1/133a could be temporarily inactivated by genetic engineering. Indeed, the expression of the genes for the oncostatin M receptor and the FGF receptor 1 increased in these cells, and with it the number of receptor molecules. This triggered intracellular processes, at the end of which cell division was initiated in the cardiac muscle cells. "Conversely, overexpression of miR-1/133a caused the cells to terminate any cell division activity if we had previously induced it by administering the binding molecules of the two receptors, i.e. oncostatin and FGF," explains Melissa Valussi, first author of the study.
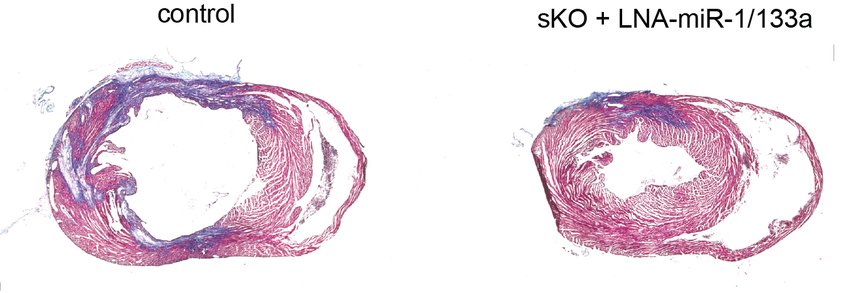
In their studies on genetically modified mice, the Bad Nauheim researchers also found that in animals in which miR-1/133a was switched off, the heart muscle cells coped much better with hypoxia conditions (oxygen depletion), such as those that exist after a heart attack. "In the experiment, the heart muscle cells from miR-1/133a knockout animals showed higher viability and survival in hypoxia," Valussi said. "In addition, we found that these animals developed a much smaller scar on the heart muscle after experimentally induced myocardial infarction. Clearly, the absence of miR-1/133a had a positive effect on myocardial cell survival and regeneration." However, this effect was reversed when miR-1/133a was absent for a longer period of time. In this case, the heart muscle cells lost the ability to contract due to the unchecked activity of the two receptors and the animals suffered heart failure.
From the Max Planck researchers' point of view, the study shows that temporarily switching off miR-1/133a is an approach to enable the regeneration of a damaged heart muscle. "With the increasing understanding of how miR-1/133a functions in heart muscle cells, starting points are emerging for a therapeutic approach that can be used to strengthen the heart's self-healing powers," says Thomas Braun, Director at the Max Planck Institute for Heart and Lung Research.