Chemistry and Renewable Energy: Generation Gap
We need to cut our use of fossil fuels, but renewable energy sources can be unreliable. Chemists could smooth out the patchy supply of sun and wind power by developing fuels and batteries that can store energy during peak times. To be deployed at national-grid level, this technology will need to unite disparate disciplines and break political, social and economic barriers.
Hydrogen could come into its own as back-up storage for the electricity grid.
To help combat climate change and wean ourselves off fossil fuels, we need to use energy more efficiently. In Germany, for example, almost one-third of the energy is lost as energy is converted from one form to another. Research that makes these processes more efficient can help us to cut carbon emissions.

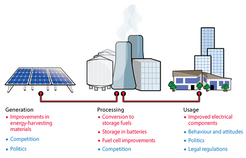
We will also need to switch to renewable electricity, however. This switch will need to be decisive in order for power generation to be both efficient and affordable, but there are further problems to overcome: wind and solar power are inherently intermittent, and at the moment energy generated from fossil fuels is used to fill the gaps. Improving energy storage would enable us to store the electricity generated at peak production times, dramatically increasing the viability of renewable energy.
Next-generation fuels and batteries do exist, but none are yet viable on an industrial scale. In the coming decade, interdisciplinary research involving chemists, materials experts and engineers will be needed to develop these innovations into real-life applications.
It’s a gas
Storing excess energy in next-generation fuels holds the most promise. The natural world has being doing this for a long time - sunlight nurtured the plants and animals that eventually formed our fossil fuels, and the energy stored by presentday plants can be used as biofuels. However, the effectiveness of existing biofuels in reducing greenhouse-gas emissions is limited - converting land to biofuel agriculture, as well as producing and transporting biofuels are carbon intensive processes1.
There are other ways of storing energy. The chemical bonds between the atoms in hydrogen gas, for example, liberate a large amount of energy when broken, making this gas very attractive for energy storage2. Hydrogen could even be generated by sunlight directly: scientists are developing ‘artificial leaves’ in which photo catalysts harness the Sun’s energy to split water into hydrogen and oxygen3. Even so, artificial leaves are still at the experimental stage, they have low overall efficiency, and their need for exotic materials is a major drawback.
Instead, hydrogen could come into its own as back-up storage in electricity grids. Excess electricity generated from renewable energy sources (Fig. 2) could be used to split water into hydrogen and oxygen through electrolysis; the energy would then be stored in the hydrogen and extracted by burning. An alternative could be to use the hydrogen to generate electricity in fuel cells, producing clean water as a by-product.
One obvious disadvantage of hydrogen gas as an energy-storage medium is that it is highly volatile - but it could be processed into ammonia4, methane or methanol, which are safer to transport. Furthermore, the electro catalytic processes involved in splitting water and combining oxygen and hydrogen are not yet stable or efficient enough to be practical, and the materials used in electrodes are prohibitively expensive. However, current electrodes could be replaced with cheaper and better alternatives, perhaps by building on existing work involving cobalt-phosphate electrodes5.
Finally, carbon dioxide itself could be a raw material for storing energy, creating an additional carbon sink in the process. Carbon dioxide is a very stable molecule, and it takes a lot of energy to collect, purify and transform it into a fuel. However, carbon-dioxide processing units could be linked to the power plants that produce the gas. Carbon dioxide could then be processed relatively easily into methane, methanol or larger molecules such as dimethyl ether that burn cleanly in combustion engines.
A better battery
Batteries also have a role to play in the switch to alternative energy systems (Fig. 1), and the redox-flow battery, which stores energy in electrolyte fluid, is of particular promise. The fluid has a low energy density, so it needs to be stored in correspondingly large tanks, thus restricting such batteries to stationary applications. Nevertheless, redox-flow batteries have the large energy capacity that battery systems will need, and they can discharge rapidly6.
Lithium-ion batteries have a much higher energy density than redox-flow batteries, and they have already proven their suitability for mobile applications such as electric vehicles. However, the driving range of electric vehicles is limited in comparison to those powered by fossil fuels, so a substantial increase in performance is needed. A key problem is that the cathode of a lithium-ion battery must do two important jobs: electrical conduction and ion storage. Separating these two functions, such as by using new carbon materials for electrical conduction and a stable anionic structure for lithium -ion storage, might offer a considerable advantage. Metal-air batteries offer even higher energy densities than lithium-ion batteries7. They work by drawing oxygen from air and then using it to oxidize a metal, generating an electrical current in the process. But metal-air batteries are plagued by problems, such as unwanted crystal growth during charge and discharge, and gradual degradation of the electrodes. Scientists will need to master the transport of ions through membranes and electrodes before these batteries see widespread use. Solid-state nuclear magnetic resonance has been useful for revealing how electrode materials behave during operation8, but more advanced analytical tools could open doors. Synchrotron-based electron spectroscopy, for example, could offer an atomic- level view of the processes that happen at the interface between solids and liquids in the battery9.
Across battery research, a common theme emerges - work is fragmented, with some scientists studying electrolytes, others electrodes and yet others the membranes that separate them. To achieve progress, those working in battery research will need to join together and take a holistic approach, connecting solid-state chemistry and electrochemistry.
Until these solutions for storing energy are sufficiently cheap and robust, it may make more sense to simply give away excess renewable electricity locally, in ways that support the overall aim of carbon reduction - at charging points for electric vehicles, for example. This reduces the need to build expensive infrastructure that would only be used for a few hours each year.
Beyond science
Making our energy system sustainable is more than just a technical challenge. The debate about renewable energy so far has often been guided by ideological preferences for one technology over another, without looking at its wider impact. We therefore need major societal, political and economic change to drive the use of novel energy-generating and -storing technologies (Fig. 2). Only with all of these factors working together can we achieve the radical change in our energy systems that we so badly need.
Excess electricity generated during peaks in production could be used to produce hydrogen, which in turn can be processed into methanol fuel using a copper catalyst. Researchers at the Fritz Haber Institute of the Max Planck Society examined this catalyst using spectroscopy, diffraction techniques and electron microscopy, and found that surface defects accounted for much of the catalytic activity (Behrens, M. et al. Science 336, 893 - 897; 2012). Their approach should enable the improvement of copper and other catalysts.













