Muscle cells need calcium ions
Structural analysis for a ryanodine receptor 1 gain insight into calcium release during muscle contraction
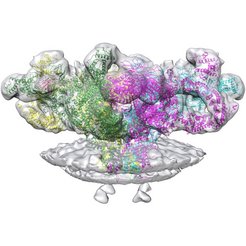
Muscle contraction is known to be regulated by calcium. An action potential generated by a motor neuron propagates on the muscle cell surface, activates voltage-gated calcium channels and allows calcium flow into the muscle cell. This calcium activates another ion channel called ryanodine receptor (RyR1 in muscle cells) which releases even more calcium stored inside the sarcoplasmic reticulum to the cytoplasm of the cell. Calcium diffusing in the cytoplasm between myosin and actin filaments of the muscle fibrils causes the filaments to slide into each other, triggering the contraction of the entire muscle fiber. As the action potential decays, the calcium ions are actively pumped back into the sarcoplasmic reticulum with the SERCAs pump (Sarcoplasmic/endoplasmic reticulum calcium ATPase).
In the current research the vesicles of sarcoplasmic reticulum were purified, imaged by cryo-electron tomography and the structure was solved by averaging to a resolution of 12.6 Å. The presence of native context showed several features which have not been observed previously; helix-like electron densities crossing the bilayer about 5 nm from the RyR1 transmembrane domain can be seen, as well as sarcoplasmic extensions connecting RyR1 with a putative calsequestrin network. The primary conformation of RyR1 in native membranes and its structural variants are documented. Surprisingly, the activation of RyR1 is associated with changes in membrane curvature and movement in the sarcoplasmic extensions.
These structures provide an insight into the mechanism of RyR1 in its natural environment.